What Does Google’s Pixel Phone Line Offer Small Business Users?
Smartphones are key communication tools in the personal and work life of…
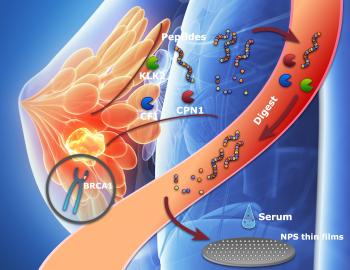
Peptides offer fresh insights for cancer diagnosis and treatment
Genes and proteins play essential roles in the maintenance of health and…
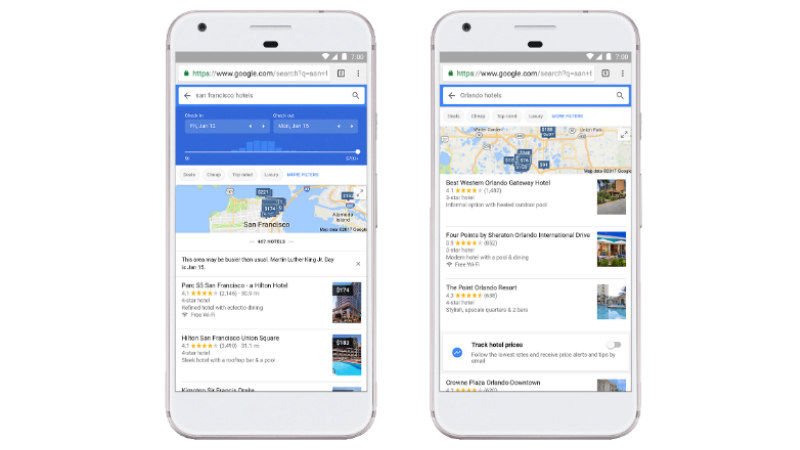
Google Flights Expands ‘Tips’ to Offer Cheaper Hotel Bookings, Google Trips Now Shows Discounts
HIGHLIGHTS Tips on Google Flights will now suggest cost-effective hotel bookings Google…
Documents Offer Insight Into Soviet View Of JFK Assassination
American Lee Harvey Oswald and his Russian wife, Marina, pose on a…